Guide to Crystallization Development
Controlling Addition Rates
With In Situ Supersaturation Profiling
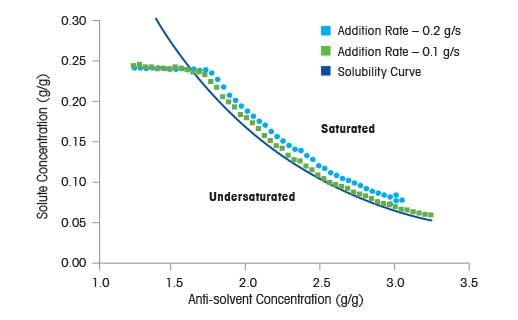
Supersaturation is generated by reducing the solubility of the product in solution, typically by cooling or adding an antisolvent. The rate at which a solution is cooled or antisolvent is added directly influences the level of supersaturation.
In this example, undersaturated solutions of benzoic acid in ethanol-water mixtures are prepared and water is added at a fixed rate of 0.1 g/s and 0.2 g/s respectively, at a fixed temperature of 25 °C. The liquid concentration is measured in real time with in situ FTIR spectroscopy. In the figure to the right, the solubility curve for benzoic acid in ethanol-water mixtures with desupersaturation profiles for each experiment are displayed. The desupersaturation profile shows that the solution begins in the undersaturated region. As water is added, the process moves past the solubility curve into the supersaturated region. The liquid concentration decreases upon crystal nucleation and stays close to the solubility curve. At the end of the antisolvent addition period, the liquid concentration drops to the solubility curve. When antisolvent is added at the faster rate, the supersaturation level is higher throughout the process - due to a buildup that cannot be relieved fast enough through crystal growth and nucleation.
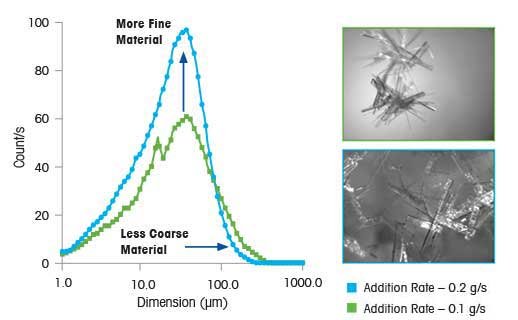
Optimize Process Parameters
To Control Supersaturation and Size
In this experiment, changing process parameters and supersaturation influence both crystal size and shape. Images captured with PVM technology (ParticleView) at the end of each experiment illustrate this point (shown on the right). The slow addition rate yields large, well-formed elongated plates, while the fast addition rate yields fine needles that readily agglomerate. This result shows that by changing supersaturation in a crystallization system, it is possible to modify crystal size, shape, and the degree of agglomeration. This also demonstrates the importance of understanding and controlling the prevailing level of supersaturation.
This simple example illustrates a critical principle:
- To make large crystals, generate supersaturation slowly
- To make small crystals, generate supersaturation quickly

Challenges with Supersaturation
Purity and Scale-up
Controlling antisolvent addition rates to control crystal size is well understood and established in sound scientific principles. However, effective and evidence-based crystallization process development and improvement is more nuanced. For example, generating supersaturation at a fast rate can lead to the generation of unwanted impurities in the form of transient oil phases (a), or unwanted polymorphic forms (b). Similarly, in an effort to generate large crystals, cycle time cannot always be sacrificed, meaning extremely slow cooling or antisolvent addition rates are not possible.

Optimizing Crystallization with Supersaturation Control
A method is presented which facilitates the calibration free use of in situ ATR-FTIR spectra for the production and control of qualitative supersaturation trajectories.
Technologies to Monitor, Optimize, and Control
Crystallization unit operations offer the unique opportunity to target and control and optimized crystal size and shape distribution to:
- Reduce Filtration and Drying Times
- Avoid Storage, Transport and Shelf Life Issues
- Ensure a Consistent and Repeatable Process at Lower Costs
Applications
Applications For Antisolvent Addition on Supersaturation
Recrystallization is a technique used to purify solid compounds by dissolving them in a hot solvent and allowing the solution to cool. During this process, the compound forms pure crystals as the solvent cools, while impurities are excluded. The crystals are then collected, washed, and dried, resulting in a purified solid product. Recrystallization is an essential method for achieving high levels of purity in solid compounds.
Solubility curves are commonly used to illustrate the relationship between solubility, temperature, and solvent type. By plotting temperature vs. solubility, scientists can create the framework needed to develop the desired crystallization process. Once an appropriate solvent is chosen, the solubility curve becomes a critical tool for the development of an effective crystallization process.
Supersaturation occurs when a solution contains more solute than should be possible thermodynamically, given the conditions of the system. Supersaturation is considered a major driver for crystallization
In-process probe-based technologies are applied to track particle size and shape changes at full concentration with no dilution or extraction necessary. By tracking the rate and degree of change to particles and crystals in real time, the correct process parameters for crystallization performance can be optimized.
Seeding is one of the most critical steps in optimizing crystallization behavior. When designing a seeding strategy, parameters such as seed size, seed loading (mass), and seed addition temperature must be considered. These parameters are generally optimized based on process kinetics and the desired final particle properties, and must remain consistent during scale-up and technology transfer.
Liquid-Liquid phase separation, or oiling out, is an often difficult to detect particle mechanism that can occur during crystallization processes.
In an antisolvent crystallization, the solvent addition rate, addition location and mixing impact local supersaturation in a vessel or pipeline. Scientists and engineers modify crystal size and count by adjusting antisolvent addition protocol and the level of supersaturation.
Crystallization kinetics are characterized in terms of two dominant processes, nucleation kinetics and growth kinetics, occurring during crystallization from solution. Nucleation kinetics describe the rate of formation of a stable nuclei. Growth kinetics define the rate at which a stable nuclei grows to a macroscopic crystal. Advanced techniques offer temperature control to modify supersaturation and crystal size and shape.
Changing the scale or mixing conditions in a crystallizer can directly impact the kinetics of the crystallization process and the final crystal size. Heat and mass transfer effects are important to consider for cooling and antisolvent systems respectively, where temperature or concentration gradients can produce inhomogeneity in the prevailing level of supersaturation.
Crystal polymorphism describes the ability of one chemical compound to crystallize in multiple unit cell configurations, which often show different physical properties.
Protein crystallization is the act and method of creating structured, ordered lattices for often-complex macromolecules.
Lactose crystallization is an industrial practice to separate lactose from whey solutions via controlled crystallization.
The MSMPR (Mixed Suspension Mixed Product Removal) crystallizer is a type of crystallizer used in industrial processes to produce high-purity crystals.
A well-designed batch crystallization process is one that can be scaled successfully to production scale - giving the desired crystal size distribution, yield, form and purity. Batch crystallization optimization requires maintaining adequate control of the crystallizer temperature (or solvent composition).
Continuous crystallization is made possible by advances in process modeling and crystallizer design, which leverage the ability to control crystal size distribution in real time by directly monitoring the crystal population.
Recrystallization is a technique used to purify solid compounds by dissolving them in a hot solvent and allowing the solution to cool. During this process, the compound forms pure crystals as the solvent cools, while impurities are excluded. The crystals are then collected, washed, and dried, resulting in a purified solid product. Recrystallization is an essential method for achieving high levels of purity in solid compounds.
Solubility curves are commonly used to illustrate the relationship between solubility, temperature, and solvent type. By plotting temperature vs. solubility, scientists can create the framework needed to develop the desired crystallization process. Once an appropriate solvent is chosen, the solubility curve becomes a critical tool for the development of an effective crystallization process.
In-process probe-based technologies are applied to track particle size and shape changes at full concentration with no dilution or extraction necessary. By tracking the rate and degree of change to particles and crystals in real time, the correct process parameters for crystallization performance can be optimized.
Seeding is one of the most critical steps in optimizing crystallization behavior. When designing a seeding strategy, parameters such as seed size, seed loading (mass), and seed addition temperature must be considered. These parameters are generally optimized based on process kinetics and the desired final particle properties, and must remain consistent during scale-up and technology transfer.
Crystallization kinetics are characterized in terms of two dominant processes, nucleation kinetics and growth kinetics, occurring during crystallization from solution. Nucleation kinetics describe the rate of formation of a stable nuclei. Growth kinetics define the rate at which a stable nuclei grows to a macroscopic crystal. Advanced techniques offer temperature control to modify supersaturation and crystal size and shape.
Changing the scale or mixing conditions in a crystallizer can directly impact the kinetics of the crystallization process and the final crystal size. Heat and mass transfer effects are important to consider for cooling and antisolvent systems respectively, where temperature or concentration gradients can produce inhomogeneity in the prevailing level of supersaturation.
A well-designed batch crystallization process is one that can be scaled successfully to production scale - giving the desired crystal size distribution, yield, form and purity. Batch crystallization optimization requires maintaining adequate control of the crystallizer temperature (or solvent composition).

















